This article was written by Ann O'Neill, RN, MBA/HCM, of Regent Surgical Health.
Ambulatory surgery centers began the mandated reporting of clinical quality indicators for individual Medicare patients through the addition of Quality Data Codes (G-Codes) to insurance claims on Oct. 1, 2012. Changes to the patient populations included in the reporting are taking effect in 2013, and new quality indicators are being added. Now is the time to strive for efficiency, reevaluate your internal processes for getting the data from the bedside to Medicare, and avoid the prospective penalties.
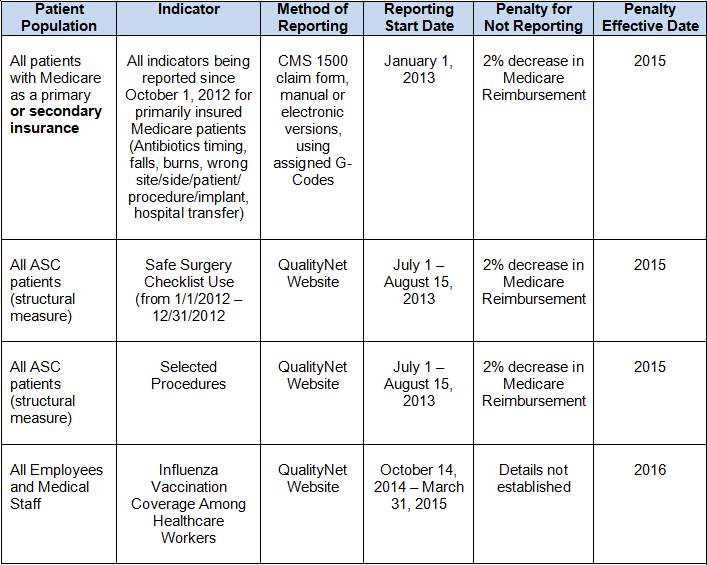
The chart below describes the who, what, and when of 2013 and 2014 reporting requirements:
ASCs have been formally reporting quality indicators to Medicare on individual claim forms for almost four months. Now is a good time to reevaluate your internal process for getting the data from the bedside to the biller and to look for opportunities to be more efficient. Collaboration between the clinical staff and the business office is essential.
One of the following methods of indicator documentation may work for you:
1. Create a one page checklist with two columns, listing the Quality Indicator in the first column and the G-code in the second column. Attach this sheet to the medical record. Instruct your RNs to circle the appropriate G-Codes in the OR and at patient discharge, as they are noted.
2. Add the indicators to your Safe Surgical Checklist (if it is in a single sheet format, and not disbursed throughout the medical record).
Entering the data into your business/billing software as close to real time as is feasible, by the clinical staff (if possible), is ideal. Attention should be paid to minimizing the number of people involved in handling and transcribing the data. These efforts will help streamline your process and improve data accuracy. A nurse who has just discharged a patient, and knows which indicators apply, can enter the data into a computer in a matter of seconds. A business office person who has to perform a chart review to extract the data may take up to five minutes per record with a completed Quality Indicator Checklist, or 10 minutes if there is no checklist.
Reporting of structural indicators, where the entire ASC patient population is included in the volume, will be performed online through www.qualitynet.org beginning July 1, 2013. ASCs will need to register at the site to create an account. This should not be done sooner than mid-March, as accounts with no activity for greater than 120 days will be automatically deleted. It is beneficial to visit the QualityNet website now to become familiar with the site's features and functions.
When anticipating the structural or facility volume indicator reporting that is starting later this year, think again about the best methods for collecting the data and who will be submitting the data through QualityNet. Use of the Safe Surgery Checklist should be second nature by now and is easily measured as it is a yes or no question. Surgical procedure volume reporting is more complex, as many procedures are included. See "Centers for Medicare & Medicaid Services Ambulatory Surgical Center Quality Reporting Program Quality Measures Specifications Manual Version 2.0, Updated: December 2012, pp22-23," for a complete list of procedures. Plan ahead and practice how you will determine and report the number of patients receiving each of these procedures, within any given timeframe, using your software or manual tracking system.
After your systems are in place for collecting and submitting quality related data, you must test the accuracy of your data to ensure compliance. Periodic tracking and measuring of data submissions for accuracy will help identify glitches in your process or data systems.
Measuring quality is natural for most ASC administrators and directors of nursing. Sharing that data with the outside world is not so natural. The day has come where we have to be transparent to both our internal and external customers. The mandated reporting of Quality Indicators to CMS is here to stay. CMS plans to add more Quality Indicators in subsequent years. Stay aware of upcoming changes, look at your tools, identify the right people to collect and enter data, and monitor your compliance. Let us do our best to make the reporting process less onerous and more efficient for all.
Learn more about Regent Surgical Health.
Ambulatory surgery centers began the mandated reporting of clinical quality indicators for individual Medicare patients through the addition of Quality Data Codes (G-Codes) to insurance claims on Oct. 1, 2012. Changes to the patient populations included in the reporting are taking effect in 2013, and new quality indicators are being added. Now is the time to strive for efficiency, reevaluate your internal processes for getting the data from the bedside to Medicare, and avoid the prospective penalties.
The chart below describes the who, what, and when of 2013 and 2014 reporting requirements:

ASCs have been formally reporting quality indicators to Medicare on individual claim forms for almost four months. Now is a good time to reevaluate your internal process for getting the data from the bedside to the biller and to look for opportunities to be more efficient. Collaboration between the clinical staff and the business office is essential.
One of the following methods of indicator documentation may work for you:
1. Create a one page checklist with two columns, listing the Quality Indicator in the first column and the G-code in the second column. Attach this sheet to the medical record. Instruct your RNs to circle the appropriate G-Codes in the OR and at patient discharge, as they are noted.
2. Add the indicators to your Safe Surgical Checklist (if it is in a single sheet format, and not disbursed throughout the medical record).
Entering the data into your business/billing software as close to real time as is feasible, by the clinical staff (if possible), is ideal. Attention should be paid to minimizing the number of people involved in handling and transcribing the data. These efforts will help streamline your process and improve data accuracy. A nurse who has just discharged a patient, and knows which indicators apply, can enter the data into a computer in a matter of seconds. A business office person who has to perform a chart review to extract the data may take up to five minutes per record with a completed Quality Indicator Checklist, or 10 minutes if there is no checklist.
Reporting of structural indicators, where the entire ASC patient population is included in the volume, will be performed online through www.qualitynet.org beginning July 1, 2013. ASCs will need to register at the site to create an account. This should not be done sooner than mid-March, as accounts with no activity for greater than 120 days will be automatically deleted. It is beneficial to visit the QualityNet website now to become familiar with the site's features and functions.
When anticipating the structural or facility volume indicator reporting that is starting later this year, think again about the best methods for collecting the data and who will be submitting the data through QualityNet. Use of the Safe Surgery Checklist should be second nature by now and is easily measured as it is a yes or no question. Surgical procedure volume reporting is more complex, as many procedures are included. See "Centers for Medicare & Medicaid Services Ambulatory Surgical Center Quality Reporting Program Quality Measures Specifications Manual Version 2.0, Updated: December 2012, pp22-23," for a complete list of procedures. Plan ahead and practice how you will determine and report the number of patients receiving each of these procedures, within any given timeframe, using your software or manual tracking system.
After your systems are in place for collecting and submitting quality related data, you must test the accuracy of your data to ensure compliance. Periodic tracking and measuring of data submissions for accuracy will help identify glitches in your process or data systems.
Measuring quality is natural for most ASC administrators and directors of nursing. Sharing that data with the outside world is not so natural. The day has come where we have to be transparent to both our internal and external customers. The mandated reporting of Quality Indicators to CMS is here to stay. CMS plans to add more Quality Indicators in subsequent years. Stay aware of upcoming changes, look at your tools, identify the right people to collect and enter data, and monitor your compliance. Let us do our best to make the reporting process less onerous and more efficient for all.
Learn more about Regent Surgical Health.

